In a small number of early seroconverters who are still in the 'window period', the p24 antigen may become positive before antibody is detectable. With the highly sensitive HIV-1/HIV-2 enzyme immunoassay (EIA) tests currently on the market, seroconversion can be detected within two to three weeks of infection in the majority of cases. Antibody testing is the method most commonly used to diagnose HIV infection. HIV infection is identified either by the detection of HIV-specific antibodies in serum or plasma or by demonstrating the presence of the virus by nucleic acid detection using polymerase chain reaction (PCR), p24 antigen testing or, rarely these days, by growing virus in cell culture. Laboratory quality assurance programs and the participation in HIV proficiency testing programs are essential to ensure that diagnostic laboratories provide accurate, timely and clinically relevant laboratory results. However, the performance of these tests requires adherence to good laboratory quality control practices, as well as the backup of a licensed diagnostic laboratory to provide confirmation and resolution of positive or indeterminate results. These tests provide rapid, on-site HIV results in a format that is relatively easy for clinic staff to perform. Point of Care tests have become increasingly popular in the United States and some places in Canada over the past several years. Laboratories should also provide specific information on specimen collection, storage and transport so that specimen integrity is not compromised, thereby preserving the accuracy of laboratory results. The choice of assays is guided by the initial screening results and the clinical information provided by the physician both are integral to the laboratory's ability to provide an accurate laboratory diagnosis. 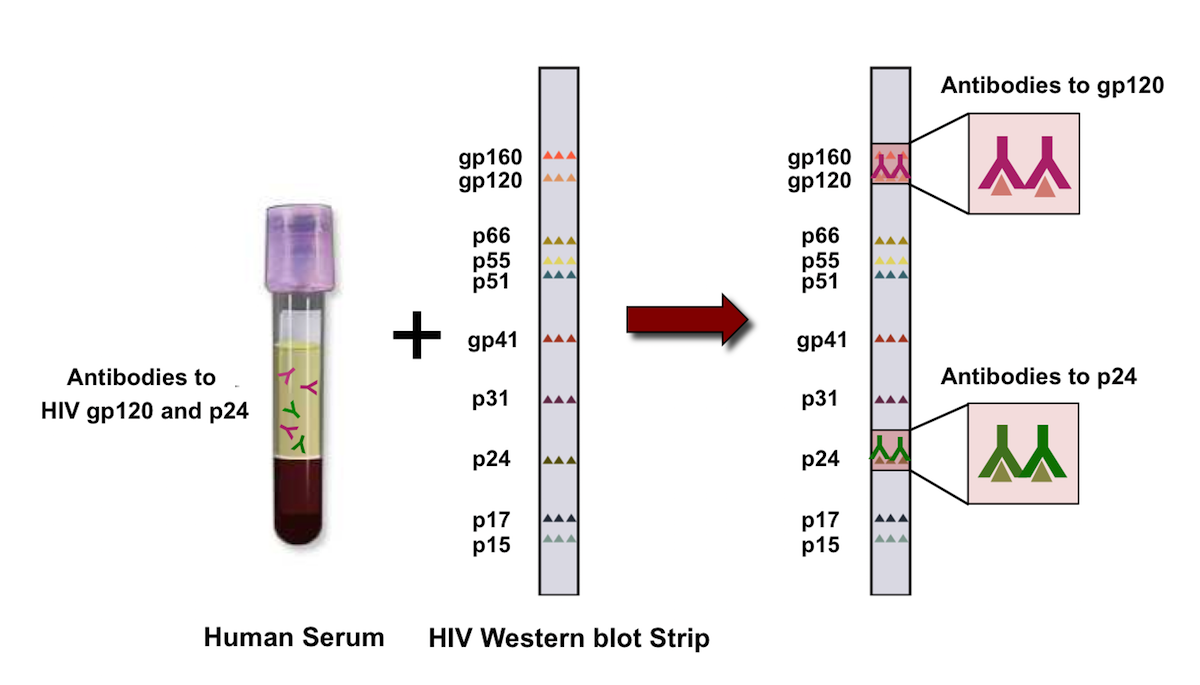
Most diagnostic laboratories have complex testing algorithms to ensure accuracy of results and optimal use of laboratory resources. A variety of other assays are essential to confirm positive antibody screens (Western blot, polymerase chain reaction ), provide an adjunct to antibody testing (p24 antigen, PCR), or provide additional information for the clinician treating HIV-positive patients (qualitative and quantitative PCR, and genotyping). Current enzyme immunoassays are sensitive enough to detect antibody as early as one to two weeks after infection. 
As well, run elisa or similar test to determine total protein content.HIV diagnostic testing has come a long way since its inception in the early 1980s. We would like to run western blots (gel filtration) to determine conjugated and unconjugated proteins. Plasma testing of 250 plasma samples from separated from human blood after performance of resistance exercise. East Coast USA Clinical Laboratory needed for ELISA and western blot testing for conjugated and unconjugated proteins. 
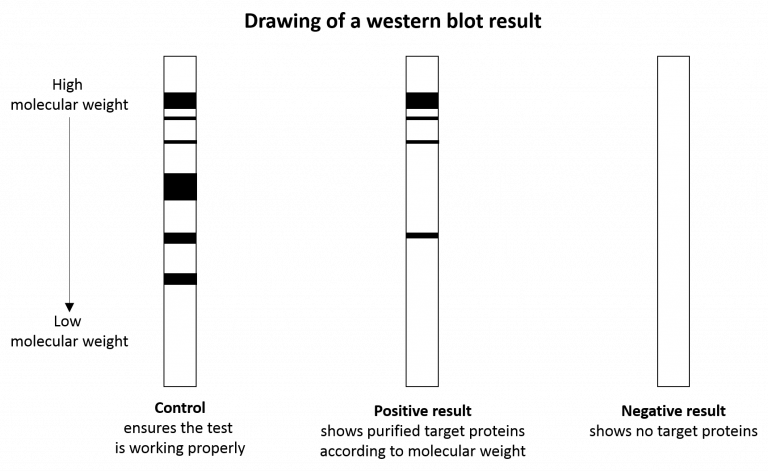
United Kingdom Bioanalytical Laboratory needed for Western Blotting and cell culture based assays.Western blotting would require probing for 5 different markers, for concentrations and incubation times required for each antibody The desired experiments would involve <24 hours treatment times. The cell culture would require the Jeg-3, JAR or BeWo cell lin Bioanalytical laboratory needed for Cell culture and Western blotting experiments.Bioanalytical laboratory needed for Western blot analysis on master reference equine horse serum to ensure that it is still highly reactive with antigen at a span of dilutions.US Bioanalytical laboratory needed for Western blot analysis using the LI-COR IR platform.Bioanalytical laboratory needed for plant hormone testing by western blot, PCR, and HPLC for cytoknin and auzin in a broth.The following are test requests we have received from companies and organizations needing Western blot testing: Western blot test requests received by Contract Laboratory The technique is very precise and useful for not only detecting proteins but also quantifying them. Western blot test is a commonly used follow-up test for HIV, Lyme disease, Hepatitis B, and other diseases as it confirms the presence of antibodies present in each of the conditions. The proteins are separated by gel electrophoresis and the antibodies are then transferred onto a membrane where if the targeted protein is present, blots or stains will appear on the membrane as proteins are separated by size and shape. The test is performed by creating artificial antibodies that will react with specific proteins. The Western Blot Test is an immunoassay, or chemical test, used to detect certain proteins in blood or other tissue samples. The Western Blot Test is done to detect certain proteins in blood or other tissue samples. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
